News
-

New partners on board of Warsaw Health Innovation Hub
Two companies have signed cooperation agreements joining the Warsaw Health Innovation Hub. Among the partners we welcome MSD Poland and Pfizer Poland.
-

New partners join the Warsaw Health Innovation Hub
A joint initiative of the Medical Research Agency and global companies is growing. MNew partners joined the Warsaw Health Innovation Hub (WHIH). Today was also the second meeting of the WHIH Council, composed of representatives of partner companies.
-

Medical Research Agency announces competition for non-commercial clinical trials in the area of civilization diseases
The main objective of the Competition is to develop new diagnostic and therapeutic procedures in the field of civilization diseases as part of non-commercial clinical trials.
-

The first Polish Centre of Support for Paediatric Clinical Research will start operating in the "Child Health Memorial Centre" Institute
The first facility in Poland dedicated to conducting clinical trials in children and adolescents was established at the Children's Memorial Health Institute in Warsaw. Today was the grand opening of the Pediatric Clinical Research Support Center. The Paediatric Clinical Trials Support Centre was established as part of competitions announced by the Medical Research Agency for the establishment and development of Clinical Trials Support Centres.
-

PLN 100 million for innovative projects in the area of rare diseases
Over 100 million PLN will be distributed by the Agency for Medical Research for the realization of 12 projects connected to rare diseases. This will provide an opportunity to develop new treatments for conditions that remain difficult to diagnose and treat due to their rarity.
-

Clinical Research Support Centre to be established at the Medical University of Lublin
The Medical University of Lublin received nearly PLN 9. 5 million from the Medical Research Agency for the creation of the Clinical Research Support Centre. The Centre will become a part of the Polish Clinical Trials Network, which aims to increase access to innovative therapies for patients.
-

The Prime Minister and the President of MRA handed over a cheque for the creation of the Silesian Clinical Research Support Centre to the SUM Rector
Prime Minister Mateusz Morawiecki and the President of the Medical Research Agency Radosław Sierpiński handed over a symbolic cheque for the amount of PLN 9,322,552. 18 to the Rector of the Medical University of Silesia in Katowice for the creation of the Silesian Clinical Research Support Centre as part of the ABM competition for the creation and development of Clinical Research Support Centres.
-
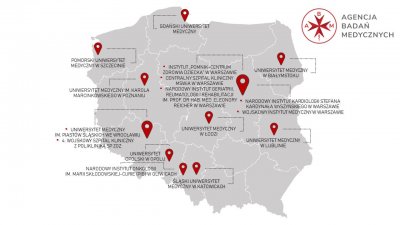
Polish Clinical Trials Network expands to include new clinical trials centers
-

Under the patronage of Prime Minister Mateusz Morawiecki, the Warsaw Health Innovation Hub was established at the Medical Research Agency.
During today's ceremony at the Chancellery of the Prime Minister, a joint project of the Medical Research Agency and global companies entitled the Warsaw Health Innovation Hub was inaugurated. This is the first initiative of its kind in Central Europe, which will combine the cooperation of the public sector with business in order to create innovative medical, technological and legal solutions for improving patients' health and increasing the efficiency of the Polish health care system.
-

Business Council of the Polish Clinical Trials Network is recruiting!
In connection with the appointment of the Polish Clinical Trials Network by Radosław Sierpiński, PhD, acting President of the Medical Research Agency, we would like to invite you to participate in the recruitment for the Business Council.