News
-

Cooperation between the Medical Research Agency and the National Science Center
President of the Medical Research Agency Radosław Sierpiński, MD, PhD and Director of the National Science Centre Professor Zbigniew Błocki signed an agreement on cooperation, including in particular mutual support in using the results of research and implementation of innovations in the health care system.
-

Medical Research Agency laureate of the Polish Intelligent Development Award 2020
Medical Research Agency has been awarded the Polish Intelligent Development Award 2020 in the category: Merit for the health of the society for actions taken to build an innovative health care system and to develop research in the field of medical and health sciences. The award ceremony will take place as part of this year's 5th edition. Intelligent Development Forum in Uniejów.
-

Medical Research Agency as the winner of Portraits of Polish Medicine 2020
Medical Research Agency has received the "Portraits of Polish Medicine" award in the category Event of the Year!
-

Medical Research Agency with the award "Innovators Wprost 2020"!
On Wednesday, 30 September, the 10th jubilee gala of "Wprost Innovators" awards was held, during which innovative products and companies as well as exceptional people were awarded for their contribution to the development of Polish science, technology and technology.
-

Results of the call or research and development activities in the field of non-commercial clinical trials - 1st round
President of the Medical Research Agency Radosław Sierpiński, MD PhD approved the ranking list containing the results of the competition for research and development activities in the field of non-commercial clinical trials in the first round of the competition.
-
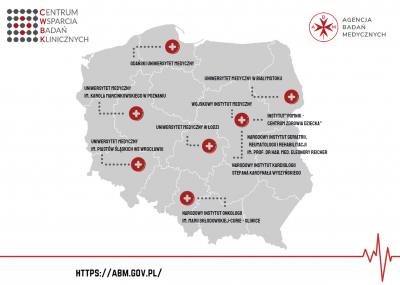
Establishment of the first network of centres conducting clinical research in Poland
The Medical Research Agency allocated PLN 100 million to create a network of ten specialised Clinical Research Support Centres. The newly established Centres will allow for better coordination of clinical trials in Poland
-

Children with lymphoblastic leukemia with a chance to be cured
Over 1100 children in Poland suffer from cancer every year. Acute lymphoblastic leukemia (ALL) is the most common malignant tumor in this group. Thanks to the financing of the Medical Research Agency, the Medical University of Lodz has started a research project CALL-POL, which will cover all children in Poland with newly diagnosed ALL.
-

MRA will donate 100 million for cancer treatment with a breakthrough CAR-T cells
The Medical Research Agency is launching a programme aimed at developing and introducing to Poland on a large scale a breakthrough cancer treatment using genetically modified CAR-T cells (adoptive therapy with CART cells). This type of therapy is particularly important in the treatment of leukaemia in the youngest patients. CAR-T cell technology changes the perspective of oncological treatment.
-

Medical Research Agency will allocate 100 million for the creation of specialized Clinical Research Support Centres.
The initiative aims to increase the number of both clinical trials and their participants in Poland, as well as to use the potential of the clinical trials sector to increase the competitiveness of the national economy.
-

The Medical Research Agency will allocate PLN 50 million to fight the coronavirus.
The Medical Research Agency and the Ministry of Health have decided to allocate at least PLN 50 million to support non-commercial clinical trials aimed at preventing COVID-19.